<<< Chapter Variants
B.1.1.7 in the US
B.1.1.7 is now spreading throughout the US. In some states, the impressive US vaccination campaign will help control the B.1.1.7-driven epidemic in the epidemic, while other states will see important surges of new cases (Figure S1). These differences in outcome will later help evaluate the usefulness of mitigation measures (Figure S2).
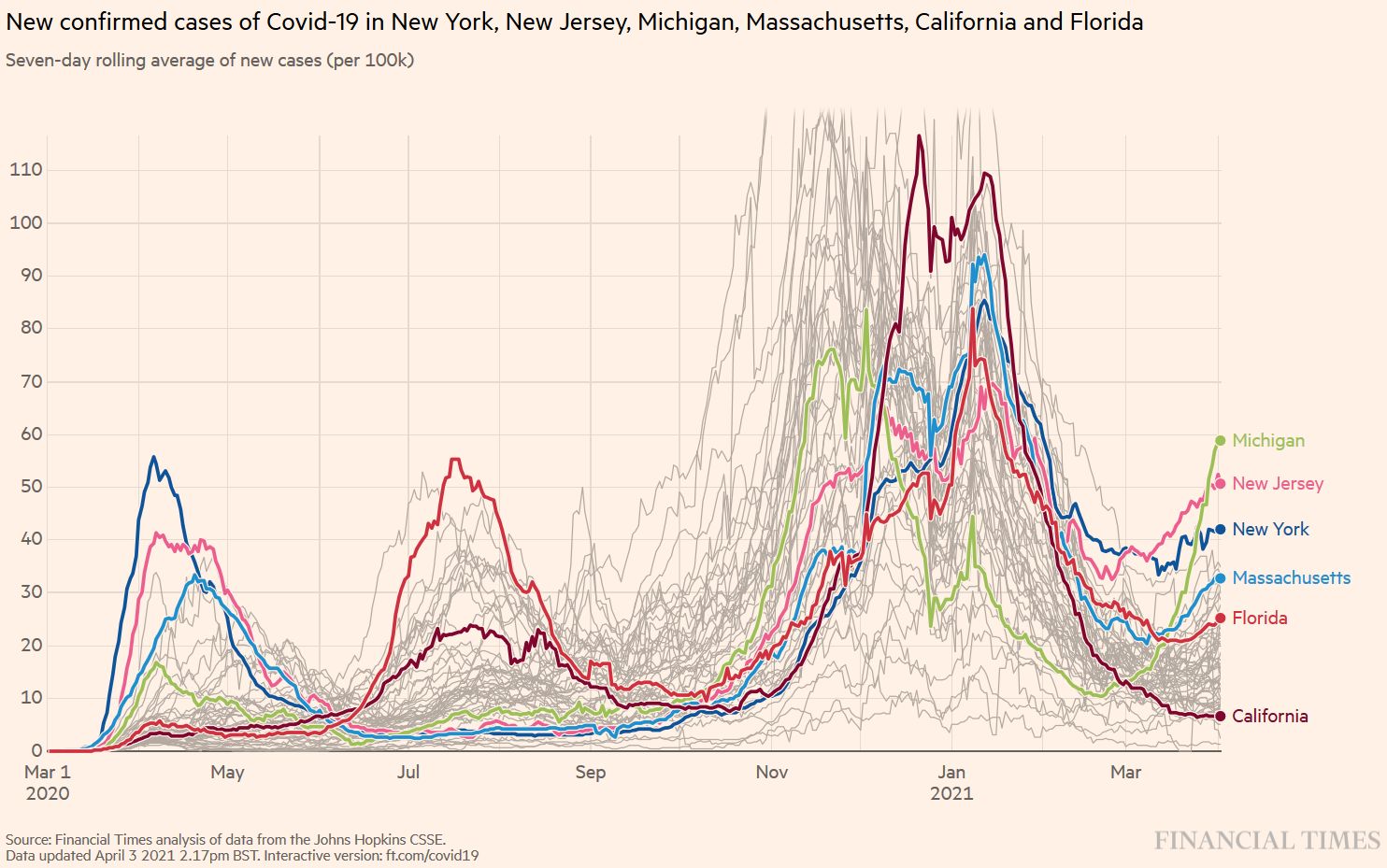
Figure S1. B.1.1.7 Spring wave in selected US states. Source and copyright: Financial Times 2021, accessed 3 April.

Figure S2. How to get out of the pandemic. Source and copyright: Wiley Miller, https://www.gocomics.com/nonsequitur, accessed 3 April.
B.1.1.7 vs B.1.351
Which variants – B.1.1.7 (first detected in England), B.1.351 (South Africa), or P.1 (Brazil) – will ultimately prevail? For now, B.1.1.7 seems to be in the lead. Data from the French Moselle department shows that in early February, the two variants from the southern hemisphere B.1.351 and P.1. accounted for 40% of new infections, compared to only 20% for B.1.1.7. One month later, B.1.1.7 was found in almost 50% of new cases (Figure S3). How long will this dominance last? Nobody knows. Once a majority of the population has been vaccinated in early autumn, the variant cards will be reshuffled.

Figure S3. Take-over of the English variant B.1.1.7 in the French Moselle department. (Souche classique: historical lineage) Source and copyright: Herzberg N. Covid-19 : qui va gagner la bataille des variants? Le Monde 2021, published 31 March. Full text: https://www.lemonde.fr/planete/article/2021/03/31/covid-19-qui-va-gagner-la-bataille-des-variants_6075043_3244.html
Important publications of the week
Transmissibility
Peng J, Liu J, Mann SA, et al. Estimation of secondary household attack rates for emergent spike L452R SARS-CoV-2 variants detected by genomic surveillance at a community-based testing site in San Francisco. Clinical Infectious Diseases 31 March 2021, ciab283, https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciab283/6206738?searchresult=1
The new lineages B.1.427 and B.1.429 (the “California” or “West Coast” variants) share S gene non-synonymous mutations at sites 13, 152, 452, and 614 and were seen during the December 2020 to February 2021 period when California was experiencing a huge peak. Whereas no instances of B.1.1.7, or independent N501Y mutations were detected in the sample population, the authors found a modest transmissibility increase of the West Coast variants. Household contacts exposed to these variants were at higher risk of infection compared to those exposed to lineages lacking these variants (0.36 vs 0.29, RR = 1.28; 95% CI: 1.00-1.64). Ct values did not differ.
Immune evasion
Dejnirattisai W, Zhou D, Supasa P, et al. Antibody evasion by the P.1 strain of SARS-CoV-2. Cell March 30, 2021. https://www.cell.com/cell/fulltext/S0092-8674(21)00428-1
All new strains (P.1 from Brazil, B.1.351 from South Africa and B.1.1.7 from the UK) have mutations in the ACE2 binding site with P.1 and B.1.351 having a virtually identical triplet: E484K, K417N/T and N501Y, conferring similar increased affinity for ACE2. Surprisingly, P.1 was significantly less resistant to naturally acquired or vaccine induced antibody responses than B.1.351, suggesting that changes outside the receptor-binding domain impact neutralization.
Ikegame S, Siddiquey MNA, Hung CT, et al. Qualitatively distinct modes of Sputnik V vaccine-neutralization escape by SARS-CoV-2 Spike variants. medRxiv 2021, posted 2 April. Full text: https://doi.org/10.1101/2021.03.31.21254660
The importance of the 2-P (proline) substitution in the spike protein in vaccines.
Vaccine efficacy
Emary KR, Gulobchik T, Aley PK. Efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine against SARS-CoV-2 variant of concern 202012/01 (B.1.1.7): an exploratory analysis of a randomised controlled trial. Lancet March 30, 2021. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(21)00628-0/fulltext
This post-hoc analysis of the efficacy of the adenoviral vector vaccine, ChAdOx1 nCoV-19 (AZD1222), revealed that laboratory virus neutralization activity by vaccine-induced antibodies was lower against B.1.1.7. However, clinical vaccine efficacy against symptomatic NAAT positive infection was good, with 70% (95% CI 44–85) for B.1.1.7 and 82% (68–89) for other lineages.
B.1.1.7
Washington NL, Gangavarapu K, Zeller M, et al. Genomic epidemiology identifies emergence and rapid transmission of SARS-CoV-2 B.1.1.7 in the United States. Cell 2021, 30 March. Full-text: https://www.cell.com/cell/fulltext/S0092-8674(21)00383-4
Kristian Andersen, Nicole L. Washington, Karthik Gangavarapu and colleagues from Helix and the Scripps Research Institute, La Jolla, sequenced 212 B.1.1.7 genomes collected in the U.S. from December 2020 to January 2021. They found a doubling rate of a little over a week and an increased transmission rate of 35-45%. The authors showed in February that the U.S. was on a similar trajectory as other countries where B117 rapidly became the dominant SARS-CoV-2 variant and warn that immediate and decisive action to minimize COVID-19 morbidity and mortality.
P.1
Francisco MA, Zavascki AP, Lamb WP, et al. Detection of SARS-CoV-2 lineage P.1 in patients from a region with exponentially increasing hospitalisation rate, February 2021, Rio Grande do Sul, Southern Brazil. Euro Surveill Accepted: 25 Mar 2021, 2021. Full text: https://doi.org/10.2807/1560-7917.ES.2021.26.12.2100276
From epidemiological week 6, starting on 7 February 2021, until 6 March, the number of hospitalizations for COVID-19 in Rio Grande do Sul, the southernmost state of Brazil in the South region, increased from 1738 inpatients to 6995 (3.8-fold). This resulted in the collapse of the state healthcare system. The overwhelming increase in hospitalizations temporally coincided with the finding that lineage P.1 became predominant (although a small number of specimens was taken).
Peer-reviewed former preprints
Cele S, Gazy I, Jackson L, et al. Escape of SARS-CoV-2 501Y.V2 from neutralization by convalescent plasma. Nature. 2021 Mar 29. PubMed: https://pubmed.gov/33780970. Full-text: https://doi.org/10.1038/s41586-021-03471-w
We already knew that people previously infected with the non-B.1.351 variant don’t neutralize B.1.351 very effectively. Now Alex Sigal, Tulio de Oliveira, Sandile Cele and colleagues show that people infected with B.1.351 can neutralize both B.1.351 and (to a slightly lesser extent) ‘regular’ non-B.1.351 viruses (Cele 2021). If these data are confirmed, a variant B.1.351-targeted booster vaccine could be a solution for countries where B.1.351 is the dominant strain.
