<<< Chapter Variants
B.1.1.7 Experiment
An informative B.1.1.7 experiment is currently taking place in Sardinia, Italy. On 26 February, the 24,090 km2 island with a population of 1.6 million was declared a ‘white zone’, allowing for the immediate expansion of various activities. Bars are expected to stay open until 9 pm, restaurants until 11 pm and the curfew may be extended from 10 pm to 11:30 pm (La Nuova Sardegna, 27 February). While all Europe is “entirely occupied by B.1.1.7, one small island of indomitable Sardinians still holds out against the invaders.” (Adapted from Astérix, 1959, by René Goscinny and Albert Uderzo.) The current epidemiological parameters (see Figure S1) would have been favorable in a pre-B.1.1.7 world, but may be less so in the presence of the B.1.1.7 variant. In Ireland, the ‘Christmas reopening’ led to a catastrophic epidemic wave (Figure S2). It will be intriguing to see if the ‘Sardinian White Zone exercise’ will be (and if so to what extent) a repeat of the Irish experience.
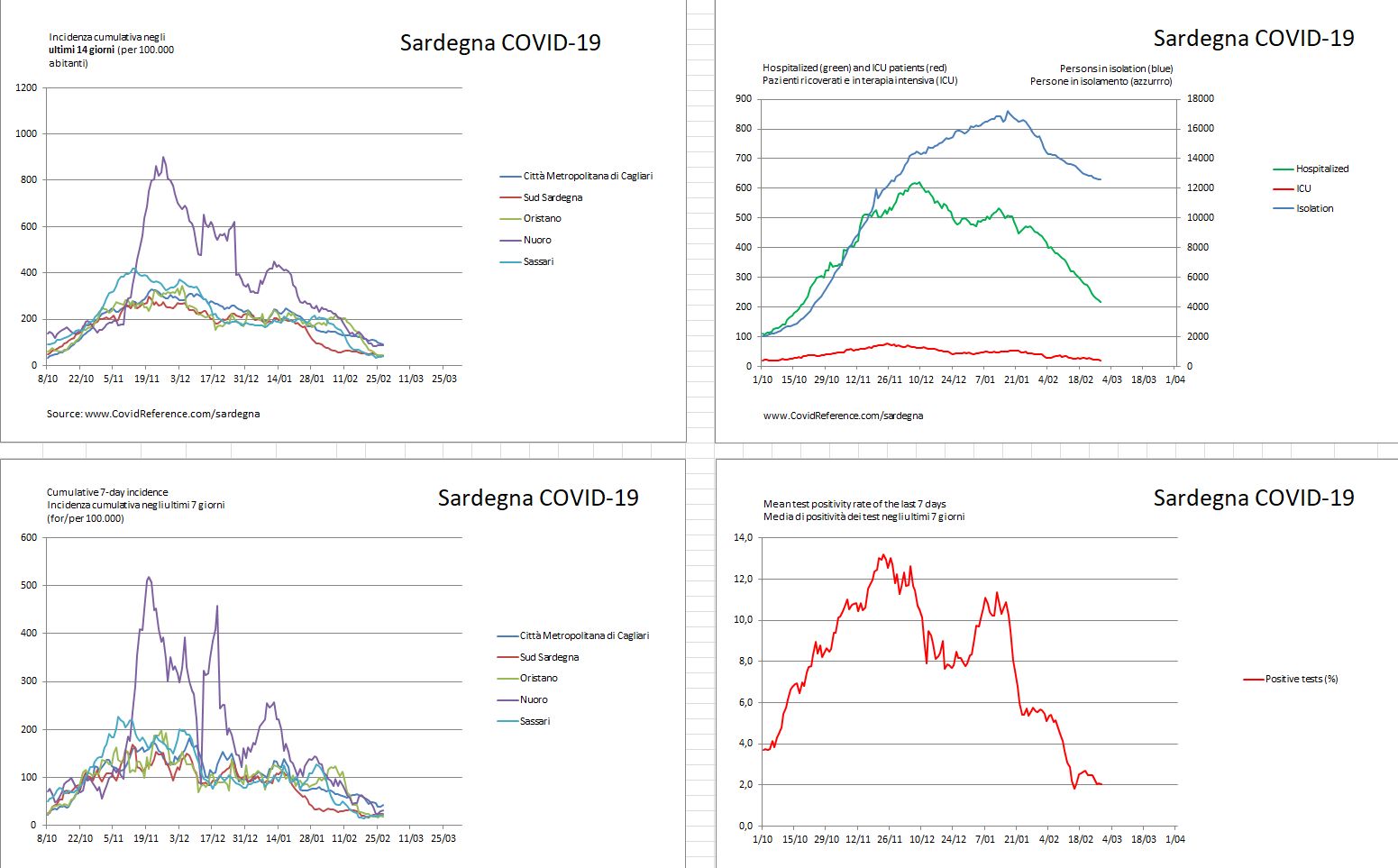
Figure S1. COVID-19 situation in Sardinia, Italy, on 27 February. (Where is Sardinia?)
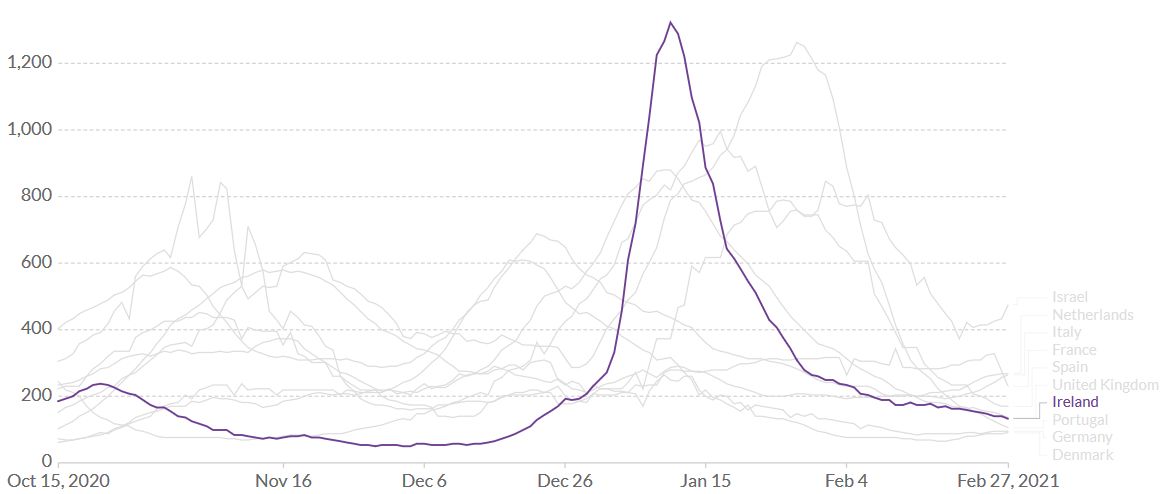
Figure S2. Ireland, January 2021. In Ireland, the proportion of B.1.1.7 was low as late as early December 2020 and Ireland seemed to be a model of a “good” pandemic response. One month later, in early January, B.1.1.7 accounted for 46% of samples tested (ECDC 20210121). The country of approximately 5 million inhabitants recorded 50.000 new SARS-CoV-2 cases in just one week and was the place where the virus was spreading the fastest in the world, with 1323 new daily cases detected per million inhabitants. The reason for the sudden resurgence is probably linked to a combination of factors: Christmas, pubs, restaurants, relaxation due to having done so well (less masks, less distancing) … plus B.1.1.7. Source: Our World in Data – Johns Hopkins University CSSE COVID-19 Data.
B.1.1.7 Dominance
The more transmissible ‘English’ variant B.1.1.7 is rapidly taking over in Europe and the US (Table S1). B.1.1.7 generally progresses from 20% to 80% of all circulating strains within four to 6 weeks (Figure S3).
| Table S1. Percentage of B.1.1.7 and B.1.351 variants in selected countries. Daily are presented as available. | ||||
| Inc.* | Percentage of B.1.1.7 | Date and source | Reaching 80%** | |
| Austria | 209 | Probably around 40-50% | ||
| Belgium | 208 | 62% | 24 Feb, Tom Wenseleers | 8 March |
| Denmark | 89 | 59.8% | 23 Feb, Danish Covid-19 Genome Consortium | 10 March |
| France | 276 | 49% | 25 Feb, Santé Publique Française | 15 March |
| Germany | 94 | 46% | 27 Feb, Anonymous (personal communication) | 15 March |
| Italy | 255 | Probably around 40-50% | ||
| Spain | 169 | No data | ||
| Switzerland | 118 | 60% | 19 Feb, Althaus et al. | 5 March |
| Florida, US | 198 | 20% | 27 Feb, Helix 202102 | April |
| UK | 143 | >90% | Already reached | |
| Ireland | 140 | >90% | Already reached | |
* 7-day incidence per 100.000 people on 26 February
** Estimate based on the Wenseleers curves: https://bit.ly/2ZV84aN + https://bit.ly/3pJP7Db
Once the proportion of B.1.1.7 of newly diagnosed SARS-CoV-2 cases exceeds 80%, a more intense epidemic activity will probably occur within 2 to 4 weeks. In Europe, a ‘normal’ Easter tourist season is highly unlikely.
As most European countries will not be able to vaccinate the adult population before the end of summer 2021, a ‘normal’ summer tourist season is equally unlikely. Countries that are unable to vaccinate well over 80% of their population (i.e., because of vaccine skepticism, etc.) will struggle to return to pre-COVID-19 normalcy.
The following Wenseleers curves describe the “One month – 20 to 80” rule of the current B.1.1.7 epidemic (Figure S1). Within one month, the percentage of the B.1.1.7 variant progresses from 20% to 80% in a given population.
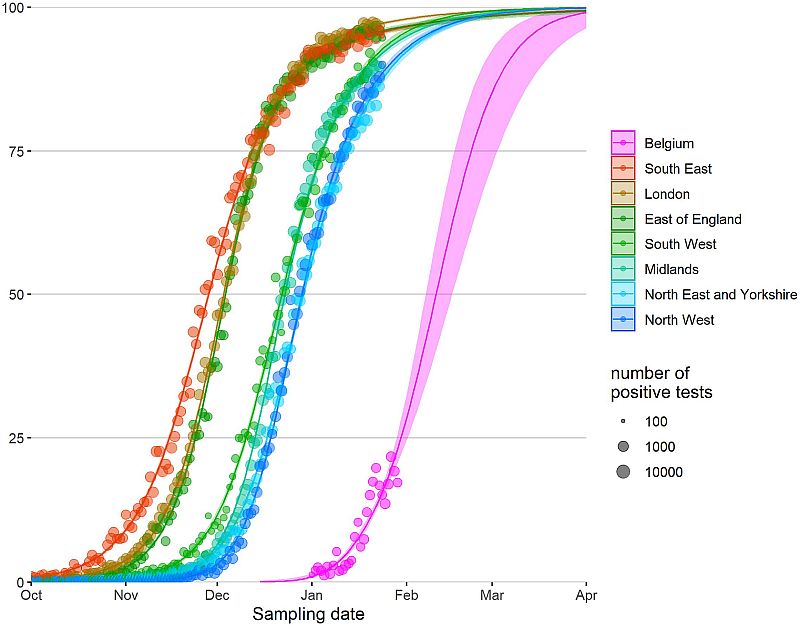
Figure S3. Relative abundance of B.1.1.7 in England and Belgium, posted on 3 February by Tom Wenseleers, https://bit.ly/3pJP7Db. The data show that the B.1.1.7 variant progressed from 20% to 80% of all circulating strains within about four weeks. Other European countries have since followed the same steep pattern.
B.1.1.7 Soft Spot
After the bad news, some good news: although B.1.1.7 is more transmissible, current vaccines retain their efficacy. Real-life data from Israel where B.1.1.7 was the dominant lineage at the beginning of the vaccination campaign in December, demonstrate that the Pfizer-BioNTech vaccine is highly effective. In a huge trial involving 596,618 vaccinees, the estimated effectiveness 7 days after the second dose was 92% for documented infection, 94% for symptomatic COVID-19, 87% for hospitalization, and 92% for severe COVID-19. Vaccine effectiveness started even two weeks after the first dose (Table S2).
| Table S2. Estimated vaccine effectiveness against COVID-19 outcomes a) 14 to 20 days after the first dose; b) 21 to 21 days after the first dose; and c) 7 days after the second dose. Adapted from Dagan N, Barda N, Kepten E, et al. BNT162b2 mRNA Covid-19 Vaccine in a Nationwide Mass Vaccination Setting. NEJM February 24, 2021. https://www.nejm.org/doi/full/10.1056/NEJMoa2101765 | |||||
| Period | Documented Infection | Symptomatic COVID-19 | Hospitalization | Severe Disease | Death |
| 14 to 20 days after first dose | 46% | 57% | 74% | 62% | 72% |
| 21 to 27 days after first dose | 60% | 66% | 78% | 80% | 84% |
| 7 days after the second dose | 92% | 94% | 87% | 92% | NA |
Also see this overview of the current leading vaccine candidates against SARS-CoV-2: Kyriakidis NC, López-Cortés A, González EV, et al. SARS-CoV-2 vaccines strategies: a comprehensive review of phase 3 candidates. npj Vaccines 6, 28 (2021). https://www.nature.com/articles/s41541-021-00292-w
